![PDF] Determination of Ammonium , Sodium , Potassium , Magnesium , and Calcium Cations in Rain Water by Column-Coupling Capillary | Semantic Scholar PDF] Determination of Ammonium , Sodium , Potassium , Magnesium , and Calcium Cations in Rain Water by Column-Coupling Capillary | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/77fdc0aa4ccba2384f1fcb3c502109ac1ddbe233/2-Table1-1.png)
PDF] Determination of Ammonium , Sodium , Potassium , Magnesium , and Calcium Cations in Rain Water by Column-Coupling Capillary | Semantic Scholar
Electronic absorbance spectra of 1.93 mmol dm −3 neptunium(IV) in 0.50... | Download Scientific Diagram

Homeostasis The control of blood sugar levels (The Regulation of Glucose in the blood) - ppt download

Basic chemical calculations. When solving numerical problems, always ask yourself whether your answer makes sense !!! - ppt download
For a given chemical reaction γ1A + γ2B → γ3C + γ4D Concentration of C changes form 10 mmol dm^-3 to 20 mmol dm^-3 - Sarthaks eConnect | Largest Online Education Community

A 5.0 m mol dm-3 aqueous solution of KCl has a conductance of 0.55 mS when measured in a cell of cell constant 1.3cm-1. The molar conductivity of this solution is _____

SOLVED: 3 A solution of concentration 0.046 mmol dm-3 records an absorbance reading of 0.893 In a dilution.5 cm3 of this solution was pipetted and then made up to 100 cm3.Calculate the

Cyclic voltammograms of 0.5 mmol dm –3 DMAFc and 1. Sweep rate, 50 mV... | Download Scientific Diagram
In a first order reaction, the concentration of reactant decreases from 20 mmol dm^-3 to 8 mmol dm^-3 in 38 minutes. - Sarthaks eConnect | Largest Online Education Community
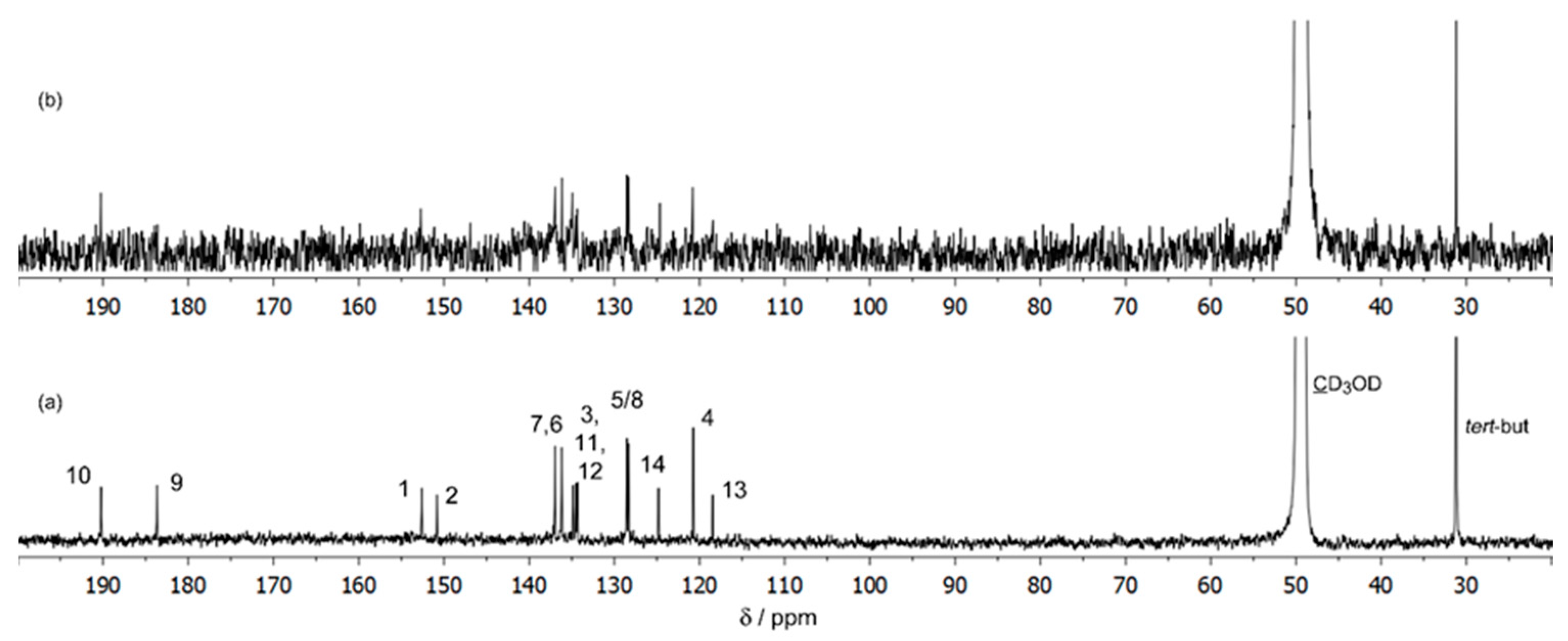
Photochem | Free Full-Text | Spectroscopic and DFT Study of Alizarin Red S Complexes of Ga(III) in Semi-Aqueous Solution
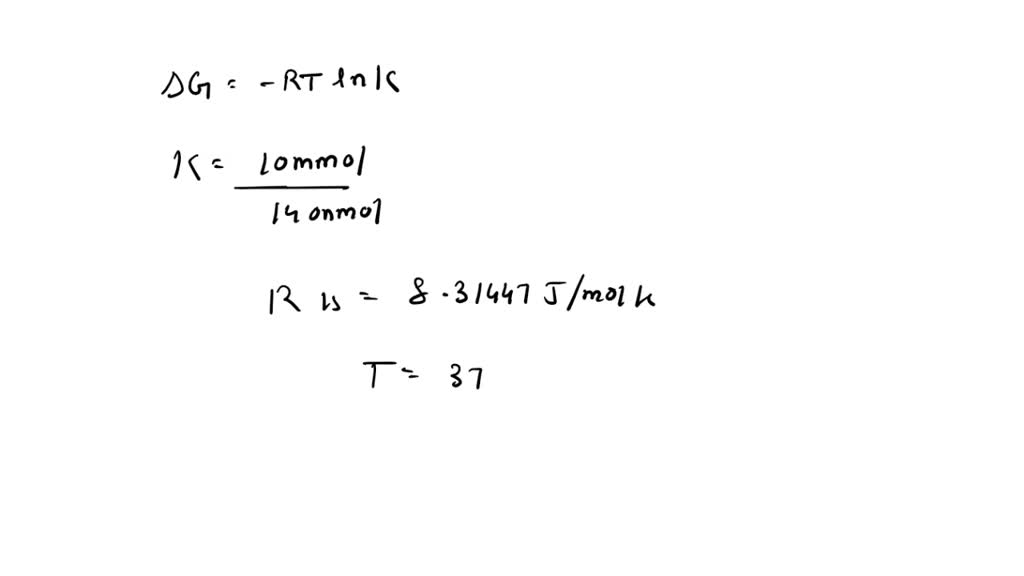
SOLVED: The distribution of Na+ ions across a typical biological membrane is 10 mmol dm-3 inside the cell and 140 mmol dm-3 outside the cell. At equilibrium, the concentrations are equal. What
A 5.0 m mol dm^–3 aqueous solution of KCl has a conductance of 0.55 mS when measured in a cell constant 1.3 cm^–1. - Sarthaks eConnect | Largest Online Education Community






![1. For the reaction A+B-C+D, a run with [A], = 400 | Chegg.com 1. For the reaction A+B-C+D, a run with [A], = 400 | Chegg.com](https://media.cheggcdn.com/media/707/707f5718-24a0-43a0-a5c7-1dc1862f2ba6/phpIjwUHj.png)




